| Cas No.: | 100286-90-6 |
| Chemical Name: | Irinotecan hydrochloride |
| Synonyms: | Irinotecan HCL;Irinotecan, Hydrochloride, Trihydrate;Irinotecan Hydrochloride;(S)-[1,4'-Bipiperidine]-1'-carboxylic acid, 4,11-diethyl-3,4,12,14-tetrahydro-4-hydroxy-3,14-dioxo-1H-pyrano[3',4':6,7]indolizino[1,2-b]quinolin-9-yl ester hydrochloride;(S)-4,11-DIETHYL-3,4,12,14-TETRAHYDRO-4-HYDROXY-3,14-DIOXO-1H-PYRANO[3',4':6,7]INDOLIZINO[1,2-B]QUINOLIN-9-YL ESTER;TOPOTECIN HYDROCHLORIDE;CPT 11;Irinotecan;Irinotecan (hydrochloride);Irinotecan HCL Trihydrate;IRINOTECAN HCL(P);IRINOTECAN MONOHYDROCHLORIDE TRIHYDRATE;Campto;Campto hydrochloride;Camptothecin 11 hydrochloride;Camptothecin analog;CPT 11 hydrochloride;CPT-11 hydrochloride;Topotecin;Camptosar;Camptothecin 11;U 101440E;06X131E4OE;NSC616348;Irinotecan hydrochloride [USAN:JAN];DSSTox_RID_81249;DSSTox_CID_25953;DSSTox_GSID_45953;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hydrochloride;Irinotecan hy... |
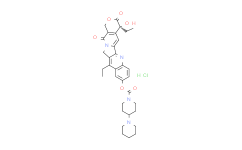
| SMILES: | Cl.CCC1C2=C(N=C3C=1C=C(C=C3)OC(=O)N4CCC(CC4)N5CCCCC5)C6N(C2)C(=O)C7=C(C=6)[C@](CC)(O)C(=O)OC7 |
| Formula: | C33H39ClN4O6 |
| M.Wt: | 623.1390 |
| Purity: | 99% |
| Sotrage: | 2 years -20°C Powder, 2 weeks 4°C in DMSO, 6 months -80°C in DMSO |
| Description: | Irinotecan hydrochloride is a water soluble topoisomerase I inhibitor mainly used to treat colon cancer and rectal cancer. |
| Target: | Topoisomerase I |
| In Vivo: | Irinotecan (CPT-11, 5 mg/kg) significantly inhibits the growth of tumors by intratumoral injection daily for 5 days, on two consecutive weeks in rats, and such effects also occur via continuous intraperitoneal infusion by osmotic minipump into mice. However, Irinotecan (10 mg/kg) shows no effect on the growth of tumor by i.p[1]. Irinotecan (CPT-11, 100-300 mg/kg, i.p.) apparently suppresses tumor growth of HT-29 xenografts in athymic female mice by day 21. The two groups of Irinotecan (125 mg/kg) plus TSP-1 (10 mg/kg per day) or Irinotecan (150 mg/kg) in combination TSP-1 (20 mg/kg per day) are nearly equally effective and inhibit tumor growth 84% and 89%, respectively, and both are more effective than Irinotecan alone at doses of 250 and 300 mg/kg[3]. |
| In Vitro: | Irinotecan hydrochloride is a topoisomerase I inhibitor. Irinotecan inhibits the growth of LoVo and HT-29 cells, with IC50s of 15.8 ± 5.1 and 5.17 ± 1.4 μM, respectively, and induces similar amounts of cleavable complexes in both in LoVo and HT-29 cells[2]. Irinotecan suppresses the proliferation of human umbilical vein endothelial cells (HUVEC), with an IC50 of 1.3 μM[3]. |
| Cell Assay: | Exponentially growing cells are seeded in 20 cm2 dishes with an optimal cell number for each cell line (20,000 for LoVo cells, 100,000 for HT-29 cells). They are treated 2 days later with increasing concentrations of irinotecan or SN-38 for one cell doubling time (24 h for LoVo cells, 40 h for HT-29 cells). After washing with 0.15 M NaCl, the cells are further grown for two doubling times in normal medium, detached from the support with trypsin-EDTA and counted in a hemocytometer. The IC50 values are then estimated as the drug concentrations responsible for 50% growth inhibition as compared with cells incubated without drug[2]. |
| Animal Administration: | Irinotecan has been administered by intratumoral injection at 0.1 cc volume of the appropriate solution, for a doses of 5 mg/kg daily for 5 days, on two consecutive weeks, followed by a 7-days rest period, referred to as one cycle of therapy. Rats receive three cycles over a period of 8 weeks. Control animals receive 0.1 cc of sterile 0.9% sodium chloride solution by intratumoral injection in the same rule of administration as that of animals of group II[1]. |
| References: | [1]. Morales C, et al. Antitumoral effect of irinotecan (CPT-11) on an experimental model of malignant neuroectodermal tumor. J Neurooncol. 2002 Feb;56(3):219-26. [2]. Pavillard V, et al. Determinants of the cytotoxicity of irinotecan in two human colorectal tumor cell lines. Cancer Chemother Pharmacol. 2002 Apr;49(4):329-35. Epub 2002 Jan 30. [3]. Allegrini G, et al. Thrombospondin-1 plus irinotecan: a novel antiangiogenic-chemotherapeutic combination that inhibits the growth of advanced human colon tumor xenografts in mice. Cancer Chemother Pharmacol. 2004 Mar;53(3):261-6. Epub 2003 Dec 5. |

 To enhance service speed and avoid tariff delays, we've opened a US warehouse. All US orders ship directly from our US facility.
To enhance service speed and avoid tariff delays, we've opened a US warehouse. All US orders ship directly from our US facility.