| Cas No.: | 71486-22-1 |
| Chemical Name: | Vinorelbine |
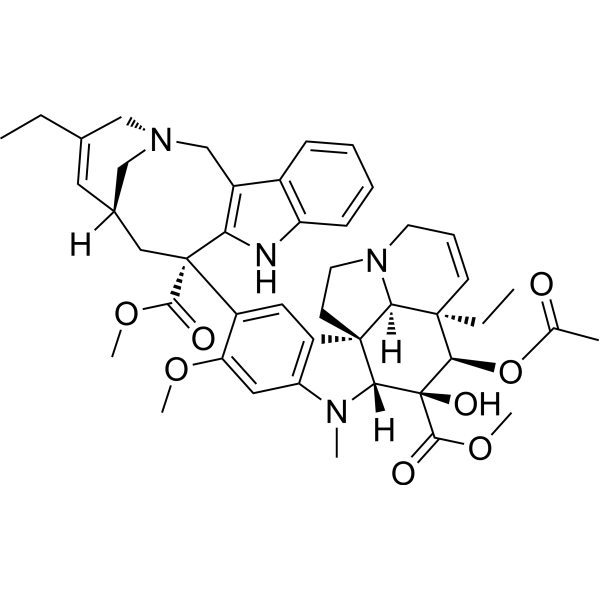
| SMILES: | CCC1=C[C@@](C[N@@]2C1)([H])C[C@@](C3=C(OC)C=C(N(C)[C@]4([H])[C@@]56[C@]7([H])[C@](C=CCN7CC6)(CC)[C@@H](OC(C)=O)[C@@]4(C(OC)=O)O)C5=C3)(C(OC)=O)C8=C(C2)C9=CC=CC=C9N8 |
| Formula: | C45H54N4O8 |
| M.Wt: | 778.94 |
| Purity: | >98% |
| Sotrage: | 4°C for 1 year, -20°C for more than 2 years |
| Description: | Vinorelbine is an anti-mitotic agent which inhibits the proliferation of Hela cells with IC50 of 1.25 nM. |
| In Vivo: | After vinorelbine treatment, the first neutropenicepisode occurred after the first (4 dogs), second (1), or sixth(1) vinorelbine treatment in the dogs[3]. Vinorelbine is tolerated at a weekly interval in tumor-bearing cats, with an MTD of 11.5 mg/m2[4]. |
| In Vitro: | Vinorelbine (0.5-5 nM) inhibits cell proliferation by 50% (IC50) at concentrations of 1.25 nM. At concentration of 8 nM vinorelbine, no cells are in anaphase[1]. Vinorelbine time-dependently induces the p53 and p21WAFI/CIP1 expression in androgen-dependent (AD) and- independent (AI) prostate cancer cell lines. Vinorelbine stimulates reporter genes in a concentration-dependent manner[2]. |
| Animal Administration: | As defined by the study, VRL1 is diluted in 0.9% NaCl to a concentration of 1.5 mg/mL, and given IV over 5 minutes. The intended treatment interval is 7 days for up to 4 treatments. After receiving 4 weekly doses, cats are eligible to continue VRL treatment every 2 weeks at the owner's expense. |
| References: | [1]. Ngan VK, et al. Mechanism of mitotic block and inhibition of cell proliferation by the semisynthetic Vinca alkaloids vinorelbine and its newer derivative vinflunine. Mol Pharmacol. 2001 Jul;60(1):225-32. [2]. Liu XM, et al. Unique induction of p21(WAF1/CIP1)expression by vinorelbine in androgen-independent prostate cancer cells. Br J Cancer. 2003 Oct 20;89(8):1566-73. [3]. Poirier VJ, et al. Toxicity, dosage, and efficacy of vinorelbine (Navelbine) in dogs with spontaneous neoplasia. J Vet Intern Med. 2004 Jul-Aug;18(4):536-9. [4]. Pierro JA, et al. Phase I clinical trial of vinorelbine in tumor-bearing cats. J Vet Intern Med. 2013 Jul-Aug;27(4):943-8. |

 To enhance service speed and avoid tariff delays, we've opened a US warehouse. All US orders ship directly from our US facility.
To enhance service speed and avoid tariff delays, we've opened a US warehouse. All US orders ship directly from our US facility.