| Cas No.: | 880635-03-0 |
| Synonyms: | GW 6471 ;GW-6471 ;GW6471 |
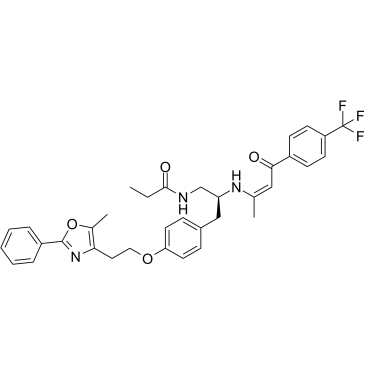
| SMILES: | CCC(NC[C@@H](N/C(C)=C\C(C1=CC=C(C(F)(F)F)C=C1)=O)CC2=CC=C(OCCC3=C(C)OC(C4=CC=CC=C4)=N3)C=C2)=O |
| Formula: | C35H36F3N3O4 |
| M.Wt: | 619.67 |
| Purity: | >98% |
| Sotrage: | 2 years -20°C Powder, 2 weeks 4°C in DMSO, 6 months -80°C in DMSO |
| Publication: | [1]. Xu HE, et al. Structural basis for antagonist-mediated recruitment of nuclear co-repressors by PPARalpha. Nature. 2002 Feb 14;415(6873):813-7. [2]. Abu Aboud O, et al. Inhibition of PPARα induces cell cycle arrest and apoptosis, and synergizes with glycolysisinhibition in kidney cancer cells. PLoS One. 2013 Aug 7;8(8):e71115. [3]. Abu Aboud O, et al. PPARα inhibition modulates multiple reprogrammed metabolic pathways in kidney cancer and attenuates tumor growth. Am J Physiol Cell Physiol. 2015 Jun 1;308(11):C890-8. |
| Description: | GW 6471 is a potent PPARα antagonist. |
| Target: | PPARα |
| In Vivo: | To test the antitumor activity of PPARα antagonism in vivo, a subcutaneous xenograft mouse model is used. Caki-1 cells are implanted subcutaneously in nude (Nu/Nu) mice. After tumor masses reach ∼5 mm in diameter, GW 6471 is administrated intraperitoneally every other day for 4 wk at a dose (20 mg/kg mouse body wt) that is described to be effective in an in vivo dose-response study and confirmed here to be efficacious. There are significant differences in tumor growth between vehicle- and GW 6471-treated animals. No toxicity is observed at the doses of GW 6471 based on weights of the animals, and laboratory values, including kidney and liver function tests, are not adversely affected. To demonstrate on-target effects of GW 6471, c-Myc levels are evaluated in the tumors, which show significant decreases in the GW 6471-treated animals[3]. |
| In Vitro: | In a cell-based reporter assay, GW 6471 completely inhibits GW409544-induced activation of PPARα with an IC50 of 0.24 μM[1]. The functional role of PPARα is evaluated on renal cell carcinoma (RCC) cell viability by MTT assay. Both Caki-1 (VHL wild type) and 786-O (VHL mutated) cells are incubated separately with a specific PPARα agonist, WY14,643, or a specific PPARα antagonist, GW 6471 at concentrations from 12.5 to 100 µM for 72 hours, and cell viability is assessed. While WY14,643 either has no affect on, or slightly increased, cell viability, GW 6471 significantly and dose-dependently inhibits cell viability (up to approximately 80%) in both cell lines[2]. |
| Cell Assay: | 786-O and Caki-1 cells are plated in 96 well plates. Both cells are incubated separately with WY14,643 or GW 6471 at concentrations from 12.5 to 100 µM for 72 hours, and after the indicated treatments, the cells are incubated in MTT solution/media mixture. Then, the MTT solution is removed and the blue crystalline precipitate in each well is dissolved in DMSO. Visible absorbance of each well at 540 nm is quantified using a microplate reader[2]. |
| Animal Administration: | Mice[3] Male athymic Nu/Nu mice (8 wk of age, ~25 g body wt) are injected with 1×105 Caki-1 cells subcutaneously (3:1 DMEM-Matrigel) in the flank region. Tumor progression is monitored weekly by calipers. When tumor size reaches ~80-100 mm3, animals are randomly assigned to four groups and treatments are started (day 1). The vehicle group receive DMSO (4% in PBS) intraperitoneally and vegetable oil via oral gavage. The PPARα group is injected intraperitoneally with GW 6471 in the same vehicle (20 mg/kg body wt; murine dose response is reported elsewhere) every other day. The Sunitinib group receive Sunitinib in vegetable oil via oral gavage (40 mg/kg body wt) 5 days/wk. Another group receive GW 6471+Sunitinib. To determine any potential toxicity of the treatment(s), body weights of the animals are measured and signs of adverse reactions are monitored. On day 28, the mice are euthanized and the tumor mass is determined. Tumor growth rate is calculated[3]. |
| References: | [1]. Xu HE, et al. Structural basis for antagonist-mediated recruitment of nuclear co-repressors by PPARalpha. Nature. 2002 Feb 14;415(6873):813-7. [2]. Abu Aboud O, et al. Inhibition of PPARα induces cell cycle arrest and apoptosis, and synergizes with glycolysisinhibition in kidney cancer cells. PLoS One. 2013 Aug 7;8(8):e71115. [3]. Abu Aboud O, et al. PPARα inhibition modulates multiple reprogrammed metabolic pathways in kidney cancer and attenuates tumor growth. Am J Physiol Cell Physiol. 2015 Jun 1;308(11):C890-8. |

 To enhance service speed and avoid tariff delays, we've opened a US warehouse. All US orders ship directly from our US facility.
To enhance service speed and avoid tariff delays, we've opened a US warehouse. All US orders ship directly from our US facility.