| Cas No.: | 191732-72-6 |
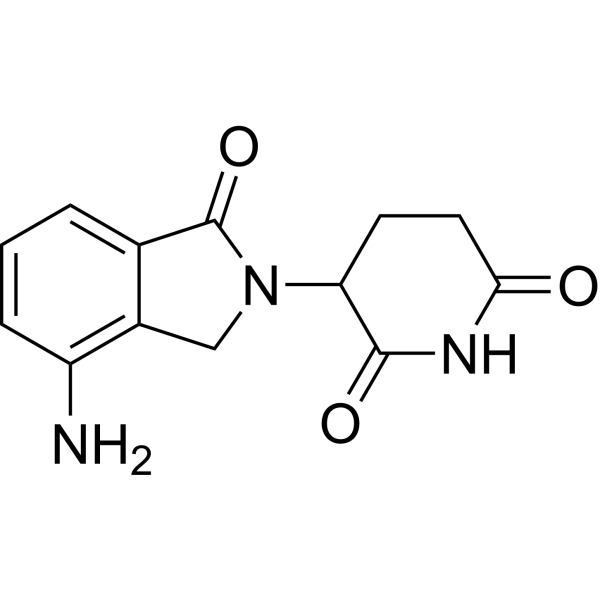
| Chemical Name: | Lenalidomide |
| Synonyms: | Lenalidomide;3-(7-amino-3-oxo-1h-isoindol-2-yl)piperidine-2,6-dione;LenalidomideLenalidomide;1-Oxo-2-(2,6-dioxopiperidin-3-yl)-4-aminoisoindoline;3-(4-Amino-1,3-dihydro-1-oxo-2H-isoindol-2-yl)-2,6-piperidinedione;CC-5013;Revlimi;Lenalidomide(other anti-cancers);(3S)-3-(4-Amino-1-oxo-1,3-dihydro-2H-isoindol-2-yl)piperidine-2,6-dione;3-(4-Amino-1-oxo-1,3-dihydro-2H-isoindol-2-yl)piperidine-2,6-dione;3-(4-Amino-1-oxo-1,3-dihydro-isoindol-2-yl)-piperidine-2,6-dione;Lenalidomide (CC-5013);Lenalidomide (CC-5013, Revlimid®); Arava;CC5013;Faslodex;Fulvestrant;HWA 486;ICI182780;Leflunomid;Leflunomida;Leflunomide;Leflunomidum;lefunamide;ZD9238;ZM182780;1-Oxo-4-amino-2-(2,6-dioxopiperidin-3-yl)isoindole;3-(4-Amino-1-oxoisoindolin-2-yl)piperidine-2,6-dione;E3 Ligase ligand;3-(4-Amino-1-oxoisoindolin-2-yl)piperidin-2,6-dione;Revlimid;To the amine;LenalidoMide-d5;LenalidoMide (RevliMid);CC 5013;Lenalidomide ,98%;SGT67 lila@tuskwei.com;Revimid;IMiD3;CDC 501;Revlimid (lenalidomide);2,6-Piperidinedione, 3-(4-amino-1,3-dihydro-1-oxo-2H-isoindol-2-yl)-;Revlimid (TN);3-(4-amino-1-oxo-2,3-dihydro-1H-isoindol-2-yl)piperidine-2,6-dione;DSSTox_RID_ |
| SMILES: | O=C1C2C([H])=C([H])C([H])=C(C=2C([H])([H])N1C1([H])C(N([H])C(C([H])([H])C1([H])[H])=O)=O)N([H])[H] |
| Formula: | C13H13N3O3 |
| M.Wt: | 259.2606 |
| Purity: | >98% |
| Sotrage: | 2 years -20°C Powder, 2 weeks 4°C in DMSO, 6 months -80°C in DMSO |
| Description: | Lenalidomide is a potent inhibitor of TNF-α used as a immunomodulatory drug. It has also been shown to have anti-angiogenic properties. |
| In Vivo: | The toxicity of Lenalidomide doses up to 15, 22.5, and 45 mg/kg via IV, IP, and PO routes of administration. Limited by solubility in our PBS dosing vehicle, these maximum achievable Lenalidomide doses are well tolerated with the exception of one mouse death (of four total dosed) at the 15 mg/kg IV dose. Notably, no other toxicities are observed in the study at IV doses of 15 mg/kg (n=3) or 10 mg/kg (n=45) or at any other dose level through IV, IP, and PO routes[4]. |
| In Vitro: | Lenalidomide is potent in stimulating T cell proliferation and IFN-γ and IL-2 production. Lenalidomide has been shown to inhibit production of pro inflammatory cytokines TNF-α, IL-1, IL-6, IL-12 and elevate the production of anti-inflammatory cytokine IL-10 from human PBMCs. Lenalidomide downregulates the production of IL-6 directly and also by inhibiting multiple myeloma (MM) cells and bone marrow stromal cells (BMSC) interaction, which augments the apoptosis of myeloma cells[2]. Dose-dependent interaction with the CRBN-DDB1 complex is observed with Thalidomide, Lenalidomide and Pomalidomide, with IC50 values of ~30 μM, ~3 μM and ~3 μM, respectively, These reduced CRBN expression cells (U266-CRBN60 and U266-CRBN75) are less responsive than the parental cells to antiproliferative effects Lenalidomide across a dose-response range of 0.01 to 10 μM[3]. Lenalidomide, a thalidomide analog, functions as a molecular glue between the human E3 ubiquitin ligase cereblon and CKIα is shown to induce the ubiquitination and degradation of this kinase, thus presumably killing leukemic cells by p53 activation[5]. |

 To enhance service speed and avoid tariff delays, we've opened a US warehouse. All US orders ship directly from our US facility.
To enhance service speed and avoid tariff delays, we've opened a US warehouse. All US orders ship directly from our US facility.