| Cas No.: | 75747-14-7 |
| Synonyms: | Tanespimycin;NSC 330507;CP 127374;17AAG;17 AAG |
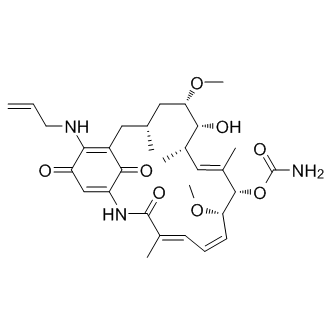
| SMILES: | O=C(C(NC(/C(C)=C/C=C[C@H](OC)[C@H](/C(C)=C/[C@@H]([C@H]([C@H](C[C@@H](C1)C)OC)O)C)OC(N)=O)=O)=CC2=O)C1=C2NCC=C |
| Formula: | C31H43N3O8 |
| M.Wt: | 585.6884 |
| Purity: | >98% |
| Sotrage: | 2 years -20°C Powder, 2 weeks 4°C in DMSO, 6 months -80°C in DMSO |
| Description: | 17-AAG is a potent HSP90 inhibitor with an IC50 of 5 nM, having a 100-fold higher binding affinity for tumour cell derived HSP90 than normal cell derived HSP90. |
| Target: | HSP90:5 nM (IC50) Mitophagy Autophagy |
| In Vivo: | 17-AAG (25-200 mg/kg, i.p.) causes a dose-dependent decline in AR, HER2, and Akt expression in prostate cancer xenografts. 17-AAG treatment at doses sufficient to induce AR, HER2, and Akt degradation results in the dose-dependent inhibition of androgen-dependent and -independent prostate cancer xenograft growth without toxicity[1]. 17-AAG (60 mg/kg) with paclitaxel (60 mg/kg) and rapamycin (30 mg/kg) inhibits A549 and MDA-MB-231 tumor growth far more potently than paclitaxel-containing micelles and effected tumor cures in MDA-MB-231 tumor-bearing animals by tail vein injection[4]. |
| In Vitro: | 17-AAG causes the degradation of HER2, Akt, and both mutant and wild-type AR and the retinoblastoma-dependent G1 growth arrest of prostate cancer cells. 17-AAG inhibits prostate cancer cell lines with IC50s ranged from 25-45 nM (LNCaP, 25 nM; LAPC-4, 40 nM; DU-145, 45 nM; and PC-3, 25 nM)[1]. Combination of 17-AAG (10 nM) and Trastuzumab induces more effective ErbB2-degradation. 17-AAG (0.1-1 μM) induces a nearly complete loss of ErbB2 on ErbB2-overexpressing breast cancer cells[2]. 17-AAG inhibits cell growth and induces G2/M cell cycle arrest and apoptosis in CCA cells together with the down-regulation of Bcl-2, Survivin and Cyclin B1, and the up-regulation of cleaved PARP[3]. |
| Cell Assay: | For the Alamar Blue proliferation assay, 2-4×103 cells are plated in 96-well plates. Later (48 h), cells are treated with 17-AAG for 96 h or 0.01% DMSO as control. On day 4, Alamar Blue viability assay is performed as described elsewhere. IC50 and IC90s are calculated as the doses of 17-AAG required to inhibit cell growth by 50 and 90%, respectively. Cell cycle distribution is assayed as described previously with a Becton Dickinson fluorescence-activated cell sorter and analyzed by the Cell Cycle Multicycle system. |
| Animal Administration: | 17-AAG is dissolved in an EPL vehicle. To aid in the identification of an optimal dose and schedule, nontumor bearing mice are treated by i.p. injection with 25-200 mg/kg of 17-AAG 5 days/week for 3 weeks or by the EPL vehicle alone. Serum samples are taken from each group, and equal volumes are pooled on days 5, 10, and 15 of treatment for serum chemistry and liver function analysis. At sacrifice, plasma samples are collected for complete blood count. A gross necropsy is performed on all of the mice, and a complete necropsy, including histopathology, is performed on 1 animal/group. |
| References: | [1]. Solit DB, et al. 17-Allylamino-17-demethoxygeldanamycin induces the degradation of androgen receptor and HER-2/neu and inhibits the growth of prostate cancer xenografts.Clin Cancer Res, 2002, 8(5), 986-993. [2]. Raja, Srikumar M., et al. A combination of Trastuzumab and 17-AAG induces enhanced ubiquitinylation and lysosomal pathway-dependent ErbB2 degradation and cytotoxicity in ErbB2-overexpressing breast cancer cells. Cancer Biology & Therapy (2008), 7(10), 163 [3]. Zhang J, et al. The heat shock protein 90 inhibitor 17-AAG suppresses growth and induces apoptosis in human cholangiocarcinoma cells.Clin Exp Med. 2012 Sep 7. [4]. Newman B, et al. HSP90 Inhibitor 17-AAG Selectively Eradicates Lymphoma Stem Cells.Cancer Res. 2012 Sep 1;72(17):4551-61. Epub 2012 Jun 29. [5]. Kamal A, et al. A high-affinity conformation of Hsp90 confers tumour selectivity on Hsp90 inhibitors. Nature. 2003 Sep 25;425(6956):407-10. |

 To enhance service speed and avoid tariff delays, we've opened a US warehouse. All US orders ship directly from our US facility.
To enhance service speed and avoid tariff delays, we've opened a US warehouse. All US orders ship directly from our US facility.