| Cas No.: | 612847-09-3 |
| Chemical Name: | Akt inhibitor VIII |
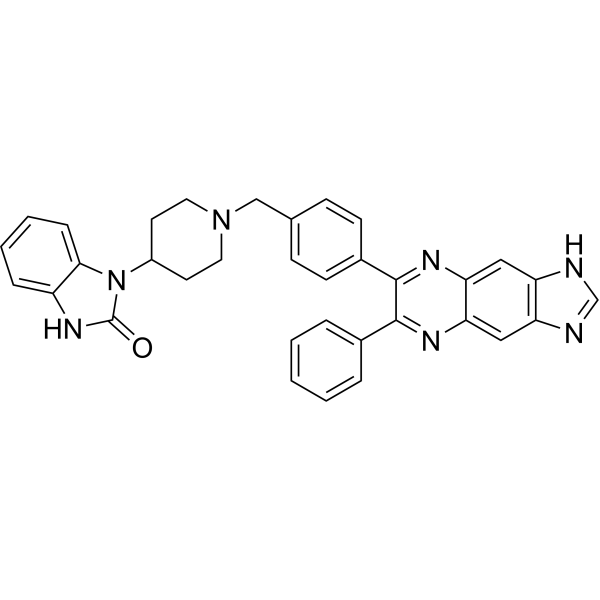
| Synonyms: | 1-(1-(4-(6-Phenyl-1H-imidazo[4,5-g]quinoxalin-7-yl)benzyl)piperidin-4-yl)-1H-benzo[d]imidazol-2(3H)-one;AKT inhibitor VIII;1-(1-(4-(6-Phenyl-1H-imidazo[4,5-g]quinoxalin-7-yl)benzyl)-piperidin-4-yl)-1H-benzo[d]imidazol-2(3H)-one;2H-Benzimidazol-2-one, 1,3-dihydro-1-[1-[[4-(6-phenyl-1H-imidazo[4,5-g]quinoxalin-7-yl)phenyl]...;2H-Benzimidazol-2-one, 1,3-dihydro-1-[1-[[4-(6-phenyl-1H-imidazo[4,5-g]quinoxalin-7-yl)phenyl]methyl]-4-piperidinyl]-;3-[1-[[4-(7-phenyl-3H-imidazo[4,5-g]quinoxalin-6-yl)phenyl]methyl]piperidin-4-yl]-1H-benzimidazol-2-one;1,3-Dihydro-1-(1-((4-(6-phenyl-1H-imidazo[4,5-g]quinoxalin-7-yl)phenyl)methyl)-4-piperidinyl)-2H-benzimidazol-2-one;Akt Inhibitor VIII, Isozyme-Selective, Akti-1;AKTI-1;imidazoquinoxaline 16h;1,3-Dihydro-1-[1-[4-(6-phenyl-1H-imidazo[4,5-g]quinoxalin-7-yl)phenylmethyl]-4-piperidinyl]-2H-benzimidazol-2-one;1,3-Dihydro-1-[1-[[4-(6-phenyl-1H-imidazo[4,5-g]quinoxalin-7-yl)phenyl]methyl]-4-piperidinyl]-2H-benzimidazol-2-one;Akti-1/2;Akt Inhibitor VIII, Isozyme-Selective, Akti-1/2;InSolution™ Akt Inhibitor VIII, Isozyme-Selective, Akti-1/2;1-{1- |
| SMILES: | O=C1N([H])C2=C([H])C([H])=C([H])C([H])=C2N1C1([H])C([H])([H])C([H])([H])N(C([H])([H])C2C([H])=C([H])C(C3=C(C4C([H])=C([H])C([H])=C([H])C=4[H])N=C4C([H])=C5C(C([H])=C4N3[H])=NC([H])=N5)=C([H])C=2[H])C([H])([H])C1([H])[H] |
| Formula: | C34H29N7O |
| M.Wt: | 551.6404 |
| Sotrage: | 2 years -20°C Powder, 2 weeks 4°C in DMSO, 6 months -80°C in DMSO |
| Description: | AKT inhibitor VIII is a cell-permeable quinoxaline compound that has been shown to potently, selectively, allosterically, and reversibly inhibit Akt1, Akt2, and Akt3 activity with IC50s of 58 nM, 210 nM, and 2119 nM, respectively. |
| In Vivo: | When LnCaP cells are pretreated with AKT inhibitor VIII and then incubated with TRAIL, a dramatic increase in caspase-3 activity (6-10-fold relative to control or TRAIL alone) is observed. This sensitization of tumor cell lines with AKT inhibitor VIII is not limited to LnCaP cells as similar apoptosis induction is observed in HT29, MCF7, and A2780 cells, among others, with chemosensitizers such as camptothecin, herceptin, and doxorubicin[1]. The furanodiene-induced decrease of p-Akt and Akt expressions is enhanced by the Akt inhibitor VIII pretreatment. Furthermore, the furanodiene-induced PARP cleavage is enhanced by Akt inhibitor VIII pretreatment. The Akt inhibitor VIII shows no effect on cleaved PARP expression but decreases the p-Akt and Akt expressions[2]. AKT inhibitor VIII decreases cell viability and increases phosphatidylserine (PS) translocation to the outer leaflet of the plasma membrane, DNA fragmentation, Caspase-9 cleavage, Caspase-3 activation and PARP proteolysis in hESC lines WA01 (H1) and WA09 (H9) and in a hiPSCs cell line generated in our laboratory (FN2.1)[3]. |
| In Vitro: | When LnCaP cells are pretreated with AKT inhibitor VIII and then incubated with TRAIL, a dramatic increase in caspase-3 activity (6-10-fold relative to control or TRAIL alone) is observed. This sensitization of tumor cell lines with AKT inhibitor VIII is not limited to LnCaP cells as similar apoptosis induction is observed in HT29, MCF7, and A2780 cells, among others, with chemosensitizers such as camptothecin, herceptin, and doxorubicin[1]. The furanodiene-induced decrease of p-Akt and Akt expressions is enhanced by the Akt inhibitor VIII pretreatment. Furthermore, the furanodiene-induced PARP cleavage is enhanced by Akt inhibitor VIII pretreatment. The Akt inhibitor VIII shows no effect on cleaved PARP expression but decreases the p-Akt and Akt expressions[2]. AKT inhibitor VIII decreases cell viability and increases phosphatidylserine (PS) translocation to the outer leaflet of the plasma membrane, DNA fragmentation, Caspase-9 cleavage, Caspase-3 activation and PARP proteolysis in hESC lines WA01 (H1) and WA09 (H9) and in a hiPSCs cell line generated in our laboratory (FN2.1)[3]. |