| Cas No.: | 66611-37-8 |
| Chemical Name: | BGP-15 HCl |
| Synonyms: | N-(2-Hydroxy-3-(piperidin-1-yl)propoxy)nicotinimidamide dihydrochloride;BGP-15 2HCl;(Z)-N'-(2-hydroxy-3-(piperidin-1-yl)propoxy)nicotiniMidaMide;N-(2-Hydroxy-3-(piperidin-1-yl)propoxy)-nicotinimidamide dihydrochloride;BGP 15;3-Pyridinecarboximidamide, N-(2-hydroxy-3-(1-piperidinyl)propoxy)-, hydrochloride (1:2);BGP 15;BGP15;s8370;SB17119;3-Pyridinecarboximidamide, N-(2-hydroxy-3-(1-piperidinyl)propoxy)-, dihydrochloride;AX8217132;(Z)-N'-(2-hydroxy-3-(piperidin-1-yl)propoxy)nicotinimidamide dihydrochloride;BGP-15 |
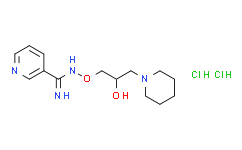
| SMILES: | Cl[H].Cl[H].O([H])C([H])(C([H])([H])O/N=C(/C1=C([H])N=C([H])C([H])=C1[H])\N([H])[H])C([H])([H])N1C([H])([H])C([H])([H])C([H])([H])C([H])([H])C1([H])[H] |
| Formula: | C14H24Cl2N4O2 |
| M.Wt: | 351.27 |
| Purity: | >98% |
| Sotrage: | 2 years -20°C Powder, 2 weeks 4°C in DMSO, 6 months -80°C in DMSO |
| Description: | BGP-15 is a PARP inhibitor, with an IC50 and a Ki of 120 and 57 μM, respectively. |
| In Vivo: | BGP-15 (15 mg/kg, p.o.) does not improve skeletal muscle pathology in older mdx mice[1]. In a rat model, 10 days of BGP-15 treatment greatly improves diaphragm muscle fiber function (by about 100%), although it does not reverse diaphragm atrophy. The treatment also provides protection from myosin PTMs associated with HSP72 induction and PARP-1 inhibition, resulting in improvement of mitochondrial function and content[2]. BGP-15 (15 mg/kg per day in saline) treatment has no effect in Ntg mice or an independent cohort of normal adult wild-type mice based on morphology, cardiac function and ECG parameters. Treatment with BGP-15 attenuates the increase in atrial size and lung weight. BGP-15 treatment is able to prevent or reduce episodes of arrhythmia. BGP-15 treatment is associated with a reduced PR interval in the HF+AF model[3]. BGP-15 (10 and 30 mg/kg) increases insulin sensitivity by 50% and 70%, respectively, in cholesterol-fed but not in normal rabbits. After 5 days of treatment with BGP-15, the glucose infusion rate is increased in a dose-dependent manner in genetically insulin-resistant GK rats. The most effective dose is 20 mg/kg, which shows a 71% increase in insulin sensitivity compared to control group[4]. |
| In Vitro: | BGP-15 (200 μM) prevents the imatinib mesylate-induced oxidative damages, attenuates the depletion of high-energy phosphates, alters the signaling effect of imatinib mesylate by preventing p38 MAP kinase and JNK activation, and induced the phosphorylation of Akt and GSK-3beta[5]. |
| Animal Administration: | Adult (appr 4 month) male HF+AF and Ntg mice are administered with BGP-15 (15 mg/kg per day in saline) for 4 weeks by oral gavage or remained untreated (oral gavage with saline or no gavage). Gavage with saline has no effect on morphological or functional parameters in the HF+AF model. Therefore, untreated mice (no gavage) and mice administered saline are combined and referred to as HF+AF control. Echocardiography and ECG studies are performed before and after treatment. |
| References: | [1]. Kennedy TL, et al. BGP-15 Improves Aspects of the Dystrophic Pathology in mdx and dko Mice with Differing Efficacies in Heart and Skeletal Muscle. Am J Pathol. 2016 Dec;186(12):3246-3260. [2]. Salah H, et al. The chaperone co-inducer BGP-15 alleviates ventilation-induced diaphragm dysfunction. Sci Transl Med. 2016 Aug 3;8(350):350ra103 [3]. Sapra G, et al. The small-molecule BGP-15 protects against heart failure and atrial fibrillation in mice. Nat Commun. 2014 Dec 9;5:5705. [4]. Literati-Nagy B, et al. Improvement of insulin sensitivity by a novel drug candidate, BGP-15, in different animal studies. Metab Syndr Relat Disord. 2014 Mar;12(2):125-31. [5]. Sarszegi Z, et al. BGP-15, a PARP-inhibitor, prevents imatinib-induced cardiotoxicity by activating Akt and suppressing JNK and p38 MAP kinases. Mol Cell Biochem. 2012 Jun;365(1-2):129-37. [6]. Szabados E, et al. BGP-15, a nicotinic amidoxime derivate protecting heart from ischemia reperfusion injury through modulation of poly(ADP-ribose) polymerase. Biochem Pharmacol. 2000 Apr 15;59(8):937-45. |

 To enhance service speed and avoid tariff delays, we've opened a US warehouse. All US orders ship directly from our US facility.
To enhance service speed and avoid tariff delays, we've opened a US warehouse. All US orders ship directly from our US facility.