| Cas No.: | 1035227-44-1 |
| Chemical Name: | AZ-505 TFA |
| Synonyms: | AZ505 (ditrifluoroacetate);N-Cyclohexyl-N3-[2-(3,4-dichlorophenyl)ethyl]-N-(2-{[2-(5-hydroxy-3-oxo-3,4-dihydro-2H-1,4-benzoxazin-8-yl)ethyl]amino}ethyl)-β-alaninamide;AZ 505 ditrifluoroacetate;AZ-505 ditrifluoroacetate;AZ-505;N-Cyclohexyl-3-[[2-(3,4-dichlorophenyl)ethyl]amino]-N-[2-[[2-(3,4-dihydro-5-hydroxy-3-oxo-2H-1,4-benzoxazin-8-yl)ethyl]amino]ethyl]propanamide 2,2,2-trifluoroacetate (1:2);AZ505 ditrifluoroacetate;N-cyclohexyl-3-((3,4-dichlorophenethyl)amino)-N-(2-((2-(5-hydroxy-3-oxo-3,4-dihydro-2H-benzo[b][1,4]oxazin-8-yl)ethyl)amino)ethyl)propanamide bis(2,2,2-trifluoroacetate);N-cyclohexyl-3-[2-(3,4-dichlorophenyl)ethylamino]-N-[2-[2-(5-hydroxy-3-oxo-4H-1,4-benzoxazin-8-yl)ethylamino]ethyl]propanamide;2,2,2-trifluoroacetic ac |
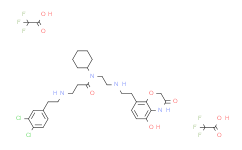
| SMILES: | ClC1=C(C([H])=C([H])C(=C1[H])C([H])([H])C([H])([H])N([H])C([H])([H])C([H])([H])C(N(C([H])([H])C([H])([H])N([H])C([H])([H])C([H])([H])C1C([H])=C([H])C(=C2C=1OC([H])([H])C(N2[H])=O)O[H])C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C1([H])[H])=O)Cl.FC(C(=O)O[H])(F)F.FC(C(=O)O[H])(F)F |
| Formula: | C29H38Cl2N4O4 |
| M.Wt: | 577.542 |
| Purity: | >98% |
| Sotrage: | 2 years -20°C Powder, 2 weeks 4°C in DMSO, 6 months -80°C in DMSO |
| Description: | AZ505 ditrifluoroacetate is a potent and selective SMYD2 inhibitor with IC50 of 0.12 μM. |
| In Vitro: | AZ505 ditrifluoroacetate is highly selective and shows an activity at submicromolar concentrations in vitro. The IC50 of AZ505 for SMYD2 is 0.12 μM, which is >600-fold greater than the IC50s of AZ505 for other histone methyltransferases, such as SMYD3 (IC50>83.3 μM), DOT1L (IC50>83.3 μM) and EZH2 (IC50>83.3 μM)[1]. AZ505 is a potent and selective SMYD2 inhibitor with an IC50 of 0.12 μM. The human SMYD (SET and MYND domain-containing protein) family of protein lysine methyltransferases contains five members (SMYD1-5). Moreover, AZ505 fails to inhibit the enzymatic activities of a panel of protein lysine methyltransferases. AZ505 is nominated for ITC binding study with Kd of 0.5 μM. In contrast, the calculated Kd for the p53 substrate peptide is 3.7 μM. AZ505 binding to SMYD2 is driven primarily by entropy, which often suggests that binding is mediated by hydrophobic interactions with few specific hydrogen bonds[2]. |
| Kinase Assay: | SMYD2 is expressed in insect cells and purified. AlphaScreen technology is used to screen our chemical library for small molecule inhibitors of SMYD2. Methylation (12 μL) reactions are carried out in TDT buffer (50 mM Tris-HCl [pH 9.0], 2 mM DTT, and 0.01% Tween 20) at room temperature using 1.25 nM SMYD2 protein, 200 nM SAM, and 100 nM biotinylated p53 peptide substrate (Biotin-aminohexanoyl-GSRAHSSHLKSKKGQSTSRH) in low volume 384-well plates. Following a 75 min incubation period, reactions are quenched by the addition of 5 μL of detection solution (20 mM HEPES [pH 7.4], 1.7 mg/mL BSA, 340 mM NaCl, 680 μM SAH, 0.04 mg/mL Streptavidin-coated AlphaScreen donor, and Protein A-coated acceptor beads), and 1 nM of a custom p53K370me1 polyclonal antibody. Reaction plates are incubated overnight in the dark at room temperature, and read using an Envision 2101 Multi-label Reader. Compounds showing >50% inhibition of SMYD2 are nominated for concentration dose-response determination, and are also subjected to an artifact assay. Seven compound concentrations are selected beginning at 30 μM with six half-log dilution steps. The artifact assay conditions are identical to those in the SMYD2 enzymatic activity assay, except for the absence of SMYD2 protein and the presence of 1 nM methylated p53 peptide. IC50 values are calculated from dose-response data using in-house software[2]. |
| References: | [1]. Komatsu S, et al. Overexpression of SMYD2 contributes to malignant outcome in gastric cancer. Br J Cancer. 2015 Jan 20;112(2):357-64. [2]. Ferguson AD, et al. Structural basis of substrate methylation and inhibition of SMYD2. Structure. 2011 Sep 7;19(9):1262-73. |

 To enhance service speed and avoid tariff delays, we've opened a US warehouse. All US orders ship directly from our US facility.
To enhance service speed and avoid tariff delays, we've opened a US warehouse. All US orders ship directly from our US facility.