| DC31074 |
Isopropyl myristate
|
Isopropyl myristate is the ester of isopropyl alcohol and myristic acid. |
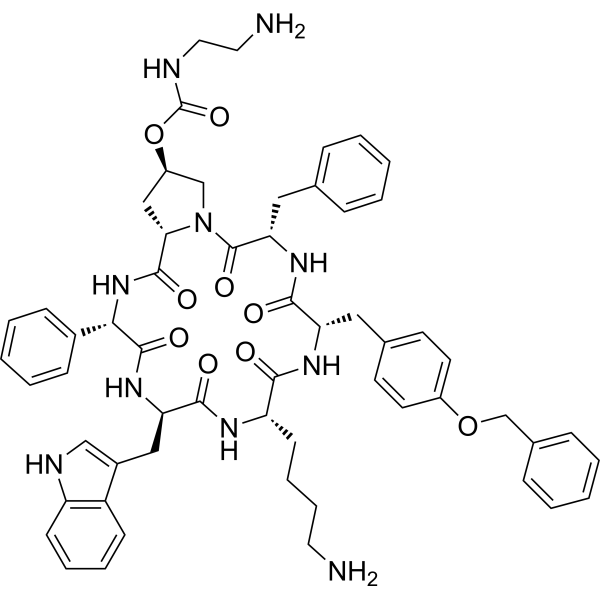
| DC75202 |
Fosaprepitant free acid
|
Fosaprepitant, also known as MK0517, is an antiemetic drug, administered intravenously. It is a prodrug of aprepitant. Fosaprepitant was developed by Merck & Co. and was approved. It is a prodrug of Aprepitant. It aids in the prevention of acute and delayed nausea and vomiting associated with chemotherapy treatment. Fosaprepitant is a weak inhibitor of CYP3A4, and aprepitant, the active moiety, is a substrate, inhibitor, and inducer of CYP3A4 |
| DC74684 |
ZH8667
|
ZH8667 is a trace amine-associated receptor 1 (TAAR1)–Gs agonist. |
| DC74646 |
EB-PSMA-617
|
EB-PSMA-617 is an Evans blue-modified prostate-specific membrane antigen (PSMA) 617 ligand for making 177Lu-EB-PSMA, which is potential useful for Metastatic Castration-Resistant Prostate Cancer. |
| DC74641 |
HC-258
|
HC-258 is a Covalent Acrylamide TEAD Inhibitor That Reduces Gene Expression and Cell Migration. HC-258 reduces the CTGF, CYR61, AXL, and NF2 transcript levels and inhibits the migration of MDA-MB-231 breast cancer cells. Co-crystallization with hTEAD2 confirmed that HC-258 binds within TEAD’s PA pocket, where it forms a covalent bond with its cysteine. |
| DC74639 |
Oligopeptide-10
|
Oligopeptide-10, also known as granactive oligopeptide-10, is a synthetic bio-active peptide composed of 15 amino acids. it can help manage acne-causing bacteria, both on its own and in conjunction with anti-acne superstar exfoliant salicylic acid. |
| DC74638 |
GLPG3667
|
GLPG3667 is an oral, reversible, and selective tyrosine kinase 2 (TYK2) inhibitor. It is being developed to treat inflammatory and auto-immune diseases. Biochemical assays showed that GLPG3667 displayed nanomolar potency on TYK2 with a selectivity over other JAK kinases >3-fold. In human PBMC, GLPG3667 showed comparable potency on the IFNα and IL-23 pathways (around 50 nM). Selectivity for TYK2 on the IFNα pathway was >14-fold and >19-fold toward the IL-2 and GM-CSF pathways in human PBMC and whole blood, respectively. Dermal ear inflammation in a mouse model of psoriasis driven by IL-23 was prevented by GLPG3667 with a minimal effective dose of 3 mg/kg given orally once daily. This effect was associated with a decrease in neutrophil infiltration and STAT3 phosphorylation at sites of inflammation. In healthy HV, GLPG3667 completely inhibited IFNα-induced STAT1 and STAT3 phosphorylation but did not impact IL-2- and GM-CSF-induced STAT5 phosphorylation. |
| DC74557 |
PREX-in1
|
PREX-in1 is a specific small-molecule inhibitor of P-Rex1 and P-Rex2 Rac-GEF activity with IC50 of 4.5 uM (P-Rex1 DHPH Rac-GEF activity) in liposome-based GEF assay, inhibits P-Rex1 and P-Rex2 through their catalytic DH domain. |
| DC74555 |
GYS32661
|
GYS32661 (GYS 32661) is a potent Rac inhibitor capable of inhibiting both Rac1 and Rac1b, inhibited activated Rac1 with IC50 of 1.18 uM in in vitro pull-down assays. |
| DC74540 |
MTX-3937
|
MTX-3937 is a small-molecule inhibitor targeting Siglec-9 (Sialic acid-binding immunoglobulin-type lectin-9), a member of the Siglec family of immunomodulatory receptors. Siglec-9 is primarily expressed on immune cells, such as myeloid cells and natural killer (NK) cells, and plays a role in regulating immune responses by transmitting inhibitory signals through its intracellular immunoreceptor tyrosine-based inhibitory motifs (ITIMs). |