| Cas No.: | 196078-29-2 |
| pH value: | Corresponds to reference standard: PASS |
| Non-reduced CE-SDS: | 98.3% |
| SEC-HPLC: | 99% |
| Isoelectric Point: | Corresponds to reference standard |
| Bacterial Endotoxins Test: | <1 EU/ml |
| Exogenous Residual DNA: | <1 pg/mg |
| Residual protein A: | <1 ng/mg |
| Biological Activity: | Compared with standard, the range ofbiological activity is 99% |
| Osmolality: | Corresponds to reference standard: PASS |
| Peptide mapping: | Corresponds to reference standard: PASS |
| N-terminal sequence: | Corresponds to reference standard:PASS |
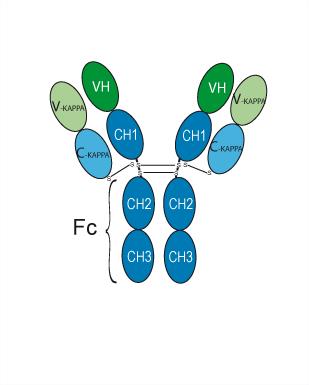
| Description: | Epratuzumab is a humanized monoclonal antibody derived from the murine IG2a monoclonal antibody, LL2 (EPB-2). |

 To enhance service speed and avoid tariff delays, we've opened a US warehouse. All US orders ship directly from our US facility.
To enhance service speed and avoid tariff delays, we've opened a US warehouse. All US orders ship directly from our US facility.