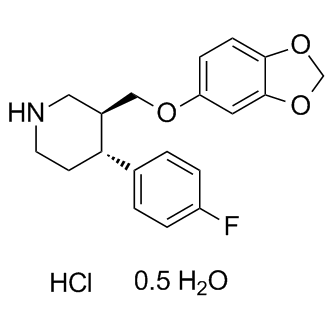
| Cas No.: | 110429-35-1 |
| SMILES: | O.FC1C=CC([C@@H]2CCNC[C@H]2COC2C=CC3OCOC=3C=2)=CC=1.FC1C=CC([C@@H]2CCNC[C@H]2COC2C=CC3OCOC=3C=2)=CC=1 |
| Formula: | C19H21ClFNO3 |
| M.Wt: | 365.83 |
| Sotrage: | 2 years -20°C Powder, 2 weeks 4°C in DMSO, 6 months -80°C in DMSO |
| Description: | Paroxetine hydrochloride hemihydrate is a potent selective serotonin-reuptake inhibitor, commonly prescribed as an antidepressant and has GRK2 inhibitory ability with IC50 of 14 μM. |
| In Vivo: | Paroxetine treatment obviously attenuates the symptoms of CIA rats. Paroxetine treatment clearly prevents the histological damage of joints and alleviates T cells infiltration into synovial tissue. Paroxetine reveals a strong effect on inhibiting CX3CL1 production in synovial tissues[1]. Paroxetine (20 mg/kg/day) reduces the myocyte cross-sectional area in rat and ROS formation in the remote myocardium. Paroxetine reduces the susceptibility to ventricular tachycardia. Paroxetine treatment following MI decreases LV remodeling and susceptibility to arrhythmias, probably by reducing ROS formation[2]. In CCI paroxetine-treated group, paroxetine (10 mg/kg, i.p.) produces hyperalgesia at days 7 and 10 (P<0.01), but a decrease in pain behavior is seen at day 14. Moreover, paroxetine (10 mg/kg) significantly attenuates tactile hypersensitivity when compared to CCI vehicle-treated group[5]. |
| In Vitro: | Paroxetine (1 μM and 10 μM) distinctly restrains T cell migration induced by CX3CL1 through inhibiting GRK2. Paroxetine inhibits GRK2 induced activation of ERK[1]. Paroxetine (10 μM) reduces pro-inflammatory cytokines in LPS-stimulated BV2 cells. Paroxetine (0-5 μM) leads to a dose-dependent inhibition on LPS-induced production of TNF-α and IL-1β in BV2 cells. Paroxetine also inhibits lipopolysaccharide (LPS)-induced nitric oxide (NO) production and inducible nitric oxide synthase (iNOS) expression in BV2 cells. Paroxetine (5 μM) blocks LPS-induced JNK activation and attenuates baseline ERK1/2 activity in BV2 cells. Paroxetine relieves microglia-mediated neurotoxicity, and suppresses LPS-stimulated pro-inflammatory cytokines and NO in primary microglial cells[4]. |

 To enhance service speed and avoid tariff delays, we've opened a US warehouse. All US orders ship directly from our US facility.
To enhance service speed and avoid tariff delays, we've opened a US warehouse. All US orders ship directly from our US facility.