| Cas No.: | 864750-70-9 |
| Synonyms: | TD 4208,TD4208 |
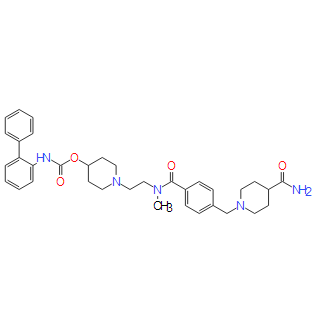
| SMILES: | C1C=C(NC(OC2CCN(CCN(C(C3C=CC(CN4CCC(C(=O)N)CC4)=CC=3)=O)C)CC2)=O)C(C2C=CC=CC=2)=CC=1 |
| Formula: | C35H43N5O4 |
| M.Wt: | 597.75 |
| Purity: | >98% |
| Sotrage: | 2 years -20°C Powder, 2 weeks 4°C in DMSO, 6 months -80°C in DMSO |
| Description: | Revefenacin (TD-4208; GSK1160724) is a potent mAChR antagonist; has a high affinity on M3 receptor with a Ki of 0.18 nM |
| Target: | Ki: 0.42 nM (M1), 0.32 nM (M2), 0.18 nM (M3), 0.56 nM (M4), 6.7 nM (M5)[1] |
| In Vivo: | In anesthetized dogs, revefenacin, along with tiotropium and glycopyrronium, produce sustained inhibition of acetylcholine-induced bronchoconstriction for up to 24 hours. In anesthetized rats, inhaled revefenacin exhibits dose-dependent 24-hour bronchoprotection against methacholine-induced bronchoconstriction. The estimated 24-hour potency is 45.0 µg/mL and the bronchoprotective potencies are maintained after 7 days of once-daily dosing[2]. |
| In Vitro: | The Kis of revefenacin are 0.42, 0.32, 0.18, 0.56, and 6.7 nM at human M1, M2, M3, M4 and M5 receptors, respectively. In a functional assay, revefenacin is shown to be a functional antagonist with inhibition constants similar to binding Ki's. Revefenacin also inhibits agonist-induced contraction of guinea pig isolated tracheal ring preparation with an affinity of 0.1 nM, similar to the measured M3 biding Ki[1]. |
| Animal Administration: | Rats: To determine the bronchoprotective and antisialagogue potency after a single dose, rats are exposed by inhalation to a nebulized solution of revefenacin (3–3000 µg/mL), tiotropium (0.3–300 µg/mL), glycopyrronium (1–1000 µg/mL), or vehicle (sterile water). Bronchoprotective activity is assessed 24 hours postdose. For the antisialagogue effect, inhibition of Pilo is assessed 1, 6, or 12 hours after inhalation of an efficacious dose of test compound to determine the time point at which peak effect occurred. All subsequent doses are measured at this time point[2]. |
| References: | [1]. Steinfeld T, et al. In vitro characterization of TD-4208, a lung-selective and long-acting muscarinic antagonist bronchodilator (Abstract). Am J Respir Crit Care Med 179:A4553. [2]. Pulido-Rios MT, et al. In vivo pharmacological characterization of TD-4208, a novel lung-selective inhaled muscarinic antagonist with sustained bronchoprotective effect in experimental animal models. J Pharmacol Exp Ther. 2013 Aug;346(2):241-50. |

 To enhance service speed and avoid tariff delays, we've opened a US warehouse. All US orders ship directly from our US facility.
To enhance service speed and avoid tariff delays, we've opened a US warehouse. All US orders ship directly from our US facility.