| Cas No.: | 302803-72-1 |
| Chemical Name: | HPI-4,Hedgehog Pathway Inhibitor 4 |
| Synonyms: | HPI-4,Hedgehog Pathway Inhibitor 4;Ciliobrevin A;3-(2,4-dichiorophenyl)-3-oxo-2-(4-oxo-3,4-dihydroquinazolin-2(1H)ylidene)propanenitrile;HPI-4;2,4-Dichloro-a-(3,4-dihydro-4-oxo-2(1H)-quinazolinylidene)-β-oxo-benzenepropanenitrile;Hedgehog Pathway Inhibitor 4;2,4-Dichloro-alpha-(3,4-dihydro-4-oxo-2(1H)-quinazolinylidene)-beta-oxobenzenepropanenitrile |
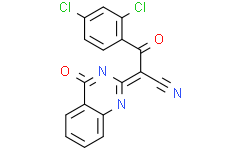
| SMILES: | N#C/C(C(C1=CC=C(Cl)C=C1Cl)=O)=C(N2)\NC3=C(C=CC=C3)C2=O |
| Formula: | C17H9N3O2Cl2 |
| M.Wt: | 358.17826 |
| Purity: | >98% |
| Description: | Ciliobrevin A is a hedgehog (Hh) signaling pathway inhibitor with median inhibitory concentration (IC50) less than 10 μM. |
| In Vitro: | Ciliobrevin A (HPI-4) also prevents an increase in the FLAG-Gli2 full-length/repressor ratio upon Shh stimulation, but HPI-2 and HPI-3 have no significant effect. Ciliobrevin A decreases FLAG-Gli1 stability in these cells, revealing another mechanism by which this small molecule can inhibit Hh target gene expression, while neither HPI-2 or HPI-3 has any significant effect on FLAG-Gli1 levels. Ciliobrevin A increases ciliary levels of FLAG-Gli2 in a manner disproportionate to their effects on total FLAG-Gli2 levels. In addition, Shh-EGFPFLAG-Gli2 cells cultured with Ciliobrevin A have truncated primary cilia, and this cellular organelle is absent in a significant fraction of Ciliobrevin A-treated cells. Ciliobrevin A also perturbs primary cilia formation in the Shh-LIGHT2FLAG-Gli1 cells and promotes accumulation of FLAG-Gli1 at the distal tip of this organelle. Ciliobrevin A significantly inhibits the proliferation of these neuronal progenitors, as measured by histone H3 phosphorylation (pH3) levels, and reduces cellular levels of cyclin D1 protein and Gli1, Gli2, and N-Myc transcripts in the CGNPs. Ciliobrevin A can block the proliferation of SmoM2-expressing CGNPs and should be equally potent against CGNPs lacking Su(fu) function, whereas the Smo inhibitor Cyclopamine is ineffective against either oncogenic lesion[1]. |
| Kinase Assay: | Smo-binding assays are conducted with BODIPY-cyclopamine and Smo-overexpressing HEK 293T cells, using a CMVpromoter-based SV40 origin-containing expression construct for Smo-Myc3 (murine Smo containing three consecutive Myc epitopes at the C terminus). HEK 293T cells are seeded into eight-well chambered coverslips (80,000 cells/well) and cultured in DMEM containing 10% FBS, 100 U/mL penicillin, and 0.1 mg/mL streptomycin. The cells are cultured until they reached 55 to 65% confluency (14-18 h), after which they are transfected with the Smo-Myc3 expression construct and Transit-LT1. Twenty-four hours after transfection, the cells are washed with PBS and cultured in DMEM containing 0.5% FBS, 5 nM BODIPY-cyclopamine, and various concentrations of either cyclopamine or individual HPIs. After 30 min, 10 μM Hoescht 33342 is added to each well, and the HPIs are incubated with the cells for an additional 30 min. The cells are then washed two times with PBS buffer, once with phenol red-free DMEM containing 0.5% FBS, and immediately imaged using a DMI6000B compound microscope. Images are background-substracted using ImageJ software with a rolling ball size of 75 pixels, and BODIPY-cyclopamine intensity is then determined using Metamorph software. Circular regions with a diameter of 300 pixels are placed over regions containing uniformly confluent cells, and the pixel intensities of approximately 20 regions from four independent images is used to determine the average BODIPY-cyclopamine levels for each experimental condition[1]. |
| Cell Assay: | NIH 3T3 cells are seeded into 24-well plates (40,000 cell/well) containing polyD-lysine-coated 12-mm glass coverslips and cultured in DMEM containing 10% CS, 100 U/mL penicillin, and 0.1 mg/mL streptomycin until they reached 85-90% confluency. The medium is changed to DMEM containing 0.5% CS, 100 U/mL penicillin, and 0.1 mg/mL streptomycin and the cells are cultured for another 12 h. The cells are then treated with either DMSO, 3 μM cyclopamine,15 μM HPI-1, 20 μM HPI-2, 30 μM HPI-3, or 30 μM Ciliobrevin A. Shh-N-conditioned medium is added to appropriate wells at a final concentration of 5%. After 12 h, the cells are fixed in 4% paraformaldehyde for 10 min at room temperature, washed three times with PBS, permeabilized for 1 min with PBS containing 0.1% Triton X-100, washed again three times with PBS, and then blocked with PBS containing 1% normal goat serum for 3 h. The coverslips are then treated with mouse anti-N-acetylated-α-tubulin (1:1,000 in blocking buffer) and rabbit anti-Smo antibody (6) (1:2,000 dilution in blocking buffer) for 2 h at room temperature and washed 3×5 min with PBS. The coverslips are incubated next with Alexa Fluor 594-conjugated goat anti-mouse IgG and Alexa Fluor 488-conjugated donkey anti-rabbit IgG antibodies (1:1,000 dilutions in blocking buffer) for 1 h at room temperature. After washes with PBS and a 5-min incubation with 4,6-diamidino-2-phenylindole (DAPI), the samples are mounted using Prolong Gold and imaged with an inverted Leica DMIRE2 laser scaning confocal microscope[1]. |
| References: | [1]. Hyman JM, et al. Small-molecule inhibitors reveal multiple strategies for Hedgehog pathway blockade. Proc Natl Acad Sci U S A. 2009 Aug 18;106(33):14132-7. |

 To enhance service speed and avoid tariff delays, we've opened a US warehouse. All US orders ship directly from our US facility.
To enhance service speed and avoid tariff delays, we've opened a US warehouse. All US orders ship directly from our US facility.