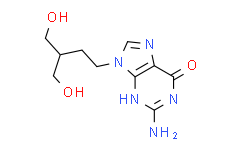
| Description: |
Penciclovir is reported to be potent against HSV types 1 and 2 with IC50 of 0.04-1.8 μg/mL and 0.06-4.4 μg/mL, respectively. |
| Target: |
IC50: 0.04-1.8 μg/mL (HSV-1), 0.06-4.4 μg/mL (HSV-2), 1.6-8 μg/mL (VZV), 1.5-3.1 μg/mL (EBV), 51 μg/mL (HCMV)[1] |
| In Vitro: |
Penciclovir inhibits herpes simplex virus type 1 (HSV-1) and type 2 (HSV-2), varicella-zoster virus (VZV), epstein-Barr virus (EBV), human cytomegalovirus (HCMV) with IC50 of 0.04-1.8 μg/mL, 0.06-4.4 μg/mL,1.6-8 μg/mL, 1.5-3.1 μg/mL, 51 μg/mL, respectively. Penciclovir (PCV) is an acyclic guanine derivative (which is not commercially available as an oral agent), has a spectrum of activity and a mechanism of action similar to those of acyclovir (ACV)[1]. |
| Cell Assay: |
Human breast cancer cell lines MCF-7 and MDA-MB-435, glioblastoma U87MG, and embryonic kidney cells 293T are cultured at 37°C in a humidified atmosphere containing 5% CO2 in Iscove’s modified Dulbecco medium or Leibovitz’s L-medium and 5% fetal bovine serum (FBS). The assays are performed with slight modifications. In brief, cells are seeded into 24-well plates (5×104 cells/well) and infected 48 h later with 103 particles per cell of unmodified virus (Adtk), PEGylated virus (PEG-Adtk), or RGD-PEG-modified virus (RGD-PEG-Adtk) in triplicates in culture medium with 2% FBS and incubated for 4 h at 37°C. The incubation medium is then replaced by normal medium and cells are further incubated for 48 h. Cells are harvested and lysed with 500 μL of TK lysis buffer that contained 0.5% Nonidet P-40 (NP-40), 20 mM N-(2-hydroxyethyl) piperazine-N′-(2-ethanesulfonic acid) (HEPES) (pH 7.6), 2 mM Mg(OAc)2, 1 mM dithiothreitol, and 50 μM thymidine. The supernatant is collected after centrifugation. The samples are kept at -80°C until use. The modified and unmodified adenovirus protein concentrations are determined by the Micro BCA assay. One microgram of cell extract is incubated with HSV1-tk substrate 8-3H-Penciclovir (8-3 H-PCV). The phosphorylated tracer is separated from unphosphorylated 8-3 H-PCV with DE-81 filters. TK activity is expressed as the percentage of conversion of substrate per minute per microgram protein[2]. |
| References: |
Human breast cancer cell lines MCF-7 and MDA-MB-435, glioblastoma U87MG, and embryonic kidney cells 293T are cultured at 37°C in a humidified atmosphere containing 5% CO2 in Iscove’s modified Dulbecco medium or Leibovitz’s L-medium and 5% fetal bovine serum (FBS). The assays are performed with slight modifications. In brief, cells are seeded into 24-well plates (5×104 cells/well) and infected 48 h later with 103 particles per cell of unmodified virus (Adtk), PEGylated virus (PEG-Adtk), or RGD-PEG-modified virus (RGD-PEG-Adtk) in triplicates in culture medium with 2% FBS and incubated for 4 h at 37°C. The incubation medium is then replaced by normal medium and cells are further incubated for 48 h. Cells are harvested and lysed with 500 μL of TK lysis buffer that contained 0.5% Nonidet P-40 (NP-40), 20 mM N-(2-hydroxyethyl) piperazine-N′-(2-ethanesulfonic acid) (HEPES) (pH 7.6), 2 mM Mg(OAc)2, 1 mM dithiothreitol, and 50 μM thymidine. The supernatant is collected after centrifugation. The samples are kept at -80°C until use. The modified and unmodified adenovirus protein concentrations are determined by the Micro BCA assay. One microgram of cell extract is incubated with HSV1-tk substrate 8-3H-Penciclovir (8-3 H-PCV). The phosphorylated tracer is separated from unphosphorylated 8-3 H-PCV with DE-81 filters. TK activity is expressed as the percentage of conversion of substrate per minute per microgram protein[2]. |