 To enhance service speed and avoid tariff delays, we've opened a US warehouse. All US orders ship directly from our US facility.
To enhance service speed and avoid tariff delays, we've opened a US warehouse. All US orders ship directly from our US facility.
| Cat. No. | Product Name | Field of Application | Chemical Structure |
|---|---|---|---|
| DC67601 | Sanofi Lipid 15 Featured |
Lipid 15, as disclosed in US Patent US 20250381150 A1 assigned to Genzyme Corporation, is an ionizable lipid used in lipid nanoparticles (LNPs) for targeted nucleic acid delivery. It features a specific structure that enables efficient encapsulation and transfection of mRNA into cells such as immune cells and hematopoietic stem cells. Experimental data show that LNPs containing Lipid 15 achieve over 80% transfection efficiency with sustained protein expression, outperforming other lipids.
More description
|

|
| DC67568 | ORNA Lipid AX-6 Featured |
AX6 is an ionizable lipid in the F32 LNP formulation, engineered by ReNAgade/Orna Therapeutics for targeted mRNA delivery to T cells. AX-6's unique bridged bicyclic/polycyclic core with a tertiary amine group enables pH-dependent protonation and endosomal escape, while C14-C18 hydrophobic tails (optionally branched/fluorinated) enhance bilayer stability and mRNA encapsulation. Demonstrating exceptional T-cell tropism, AX6 achieves high transfection efficiency in CD4+/CD8+ T cells (validated in NHP/humanized models) with minimal toxicity. Compared to clinical benchmarks (SM-102, ALC-0315), its rigid core offers superior serum stability and immune-cell specificity, positioning it as an ideal candidate for CAR-T/NK therapies and next-gen vaccines. The F32 LNP system's proven efficacy (e.g., in vivo B-cell depletion) underscores AX 6's transformative potential for cell engineering and immunotherapies.
More description
|
|
| DC60879 | Lipid te AA3-Dlin Featured |
TE AA3-Dlin is an optimized lipid nanoparticle (LNP) carrier designed for mRNA-based cancer immunotherapy, enabling precise in vivo dendritic cell (DC) reprogramming to enhance antitumor immunity. TE AA3-Dlin LNP exhibits superior serum stability, maintaining consistent particle size and low turbidity under physiological conditions, while protecting mRNA from degradation, which is crucial for effective delivery. Functionally, TE AA3-Dlin preferentially targets splenic DCs by leveraging ApoE-enriched protein coronas, facilitating efficient cellular uptake and mRNA expression, as demonstrated by enhanced EGFP signals in DCs.This targeting promotes DC maturation, antigen presentation, and membrane-bound IL-15 expression, activating cytotoxic T lymphocytes (CTLs) for tumor rejection. In models like melanoma and colon carcinoma, it synergizes with checkpoint inhibitors, showing minimal toxicity and robust immunological memory.
More description
|

|
| DC60878 | Lipid A-12 Featured |
Lipid A-12 is an ionizable cationic lipid from Capstan Therapeutics and a close analog of CICL-1 (L829). The structure was modified by the extension of the headgroup linker from a two-carbon (C2) to a three-carbon (C3) spacer compared to CICL-1 (L829).
More description
|

|
| DC67990 | 4A3-LNSC8 Featured |
4A3-LNSC8 is a strategically designed thiourea-functionalized ionizable lipid that serves as the foundational core for a novel anion-coordination delivery platform. Its structure features a central 4A3 amine headgroup symmetrically extended with four hydrophobic tails, each incorporating a biodegradable ester linkage and a key thiourea-bridged linker. The inclusion of the thiourea group is the pivotal innovation, as it provides specific hydrogen-bonding sites capable of interacting with various halide anions (F⁻, Cl⁻, I⁻). When formulated into lipid nanoparticles (LNPs) without anion coordination, 4A3-LNSC8 itself exhibits a characteristic liver tropism, efficiently delivering mRNA to hepatocytes following systemic administration, with a measured surface pKa of approximately 5.54. However, its primary significance lies in its role as a versatile precursor. The strong anion-binding capability of its thiourea linkers allows for predictable modulation of the LNP's properties. Upon binding with anions like Cl⁻, the resulting complex (e.g., Cl-4A3-LNSC8) undergoes a significant pKa shift, which reprograms the LNP's in vivo fate, redirecting mRNA delivery from the liver to secondary lymphoid organs such as the spleen and lymph nodes. Thus, 4A3-LNSC8 is not merely an efficient ionizable lipid but a programmable scaffold that enables precise control over organ-targeting specificity through simple anion coordination, offering a powerful rational design strategy for advanced mRNA therapeutics.
More description
|

|
| DC67989 | Cl-4A3-LNSC8 Featured |
Cl-4A3-LNSC8 represents a novel class of thiourea-functionalized ionizable lipids engineered for selective organ-targeted mRNA delivery. Its core innovation lies in an anion-coordination strategy, where the parent lipid, 4A3-LNSC8, binds chloride ions (Cl⁻) via hydrogen-bonding interactions with its thiourea groups. This binding event is not merely structural but functionally critical, as it induces a significant shift in the surface pKa of the resulting lipid nanoparticles (LNPs) from approximately 5.54 to 8.79. This pKa modulation is the key mechanism that redirects the organotropism of the LNPs upon systemic administration. While the unmodified 4A3-LNSC8 LNPs preferentially deliver mRNA to the liver, Cl-4A3-LNSC8 LNPs effectivelyreprogram this tropism, enabling highly efficient mRNA delivery to secondary lymphoid organs (SLOs), particularly the spleen and lymph nodes. This platform demonstrates remarkable efficacy, achieving up to 65.7% gene editing efficiency in splenic macrophages in vivo, significantly outperforming benchmark delivery systems. Furthermore, by leveraging the coordination with different halides, such as iodine for computed tomography (CT) contrast, the system can be adapted for dual-modal theranostic applications, enabling simultaneous lymphatic metastasis imaging and therapeutic mRNA delivery.
More description
|

|
| DC67602 | ILB-3132(E12LA6B603) Featured |
E12LA6B603(ILB3132,ILB-3132) is a novel ionizable amino lipid disclosed in patent WO2024198497A1, developed by MagicRNA, representing a highly efficient component for lipid nanoparticle (LNP) delivery systems.When formulated into LNPs, E12LA6B603 LNP achieves a remarkable 98.26% encapsulation efficiency for mRNA. It mediates superior in vitro transfection in dendritic cells (1.8E+05 intensity) and demonstrates best-in-class in vivo protein expression after intramuscular injection (2.2E+09 intensity). Most notably, in a B16-OVA melanoma model, therapeutic OVA-mRNA vaccines delivered by E12LA6B603 LNPs induced 100% complete tumor regression, highlighting its superior efficacy over benchmarks like DLin-MC3 and SM-102. Its biodegradable ester linkages and balanced structure make it a promising, potent candidate for next-generation mRNA vaccines and therapeutics.
More description
|

|
| DC67812 | CL15F 6-4 Featured |
CL15F 6-4 is a short-tail ionizable lipid from the piperidine-based CL15F series, characterized by its symmetric branched structure with a 6-carbon main chain and 4-carbon side chain. This specific tail length critically determines the lipid nanoparticle's (LNP) properties, resulting in larger particles with a high surface density of the phospholipid DSPC. This elevated DSPC density reduces interactions with serum proteins like ApoE, minimizing rapid liver clearance and shifting mRNA delivery preference towards the spleen. Consequently, CL15F 6-4 LNPs achieve efficient, endogenous spleen-targeted delivery, making them a highly promising candidate for enhancing vaccine efficacy by preferentially transfecting antigen-presenting cells without complex functionalization.
More description
|

|
| DC67605 | PyCB lipid Featured |
PyCB lipid (MeDZ) is a rationally designed zwitterionic ionizable lipid that serves as a core functional component in the novel three-component (ThrCo) lipid nanoparticle (LNP) platform. It is synthesized by covalently attaching a zwitterionic PyCB structure to the hydroxyl group of the clinically available ionizable lipid ALC-0315.Its key feature is its pH-responsive behavior. At physiological pH (~7.4), the PyCB headgroup exhibits zwitterionic properties, forming charge-assisted hydrogen bonds with water molecules (PyCB-H₂O complexes). This confers high hydrophilicity to the LNP surface, enhancing stability in aqueous environments and reducing nonspecific protein adsorption in the bloodstream. This zwitterionic surface effectively mimics and replaces PEGylated lipids, thereby avoiding PEG immunogenicity and the associated Accelerated Blood Clearance (ABC) effect upon repeated administrations.Crucially, in the acidic environment of endosomes (pH ~6.5), the PyCB group undergoes strong protonation, rapidly transforming into a cationic state (PyCB-H₃O⁺ complexes). This promotes efficient fusion with and disruption of the endosomal membrane, facilitating the escape and cytoplasmic release of encapsulated mRNA.By replacing both cholesterol and PEGylated lipids in traditional LNPs, PyCB lipid enables the redirection of LNP biodistribution from the liver to the spleen, achieving superior spleen-specific mRNA translation and enhancing antigen presentation for potent immune activation.
More description
|

|
| DC67450 | A28-C6B2 Featured |
A28-C6B2 is a biodegradable ionizable lipid specifically engineered for spleen-targeted delivery. Through its unique branched structure, it bypasses hepatic uptake to achieve highly efficient transfection of F4/80+ macrophages and CD11c+ dendritic cells within the spleen. This lipid remains neutral in the bloodstream to minimize non-specific interactions, while undergoing protonation in the acidic environment of the endosome to facilitate nucleic acid release, thereby significantly enhancing the potency of mRNA vaccines and immunotherapies.
More description
|

|
| DC60925 | Lipid H5T5 Featured |
H5T5 is a leading ionizable lipid nanoparticle (LNP) formulation optimized for in vivomRNA delivery, featuring a pKa of 6.51, a size of ~154 nm, and a narrow polydispersity index (PDI) of 0.05. It demonstrated superior in vitromRNA transfection efficiency in primary immune cells, such as bone marrow-derived macrophages. Following intravenous administration, H5T5 exhibits precise organotropism, predominantly targeting the spleen and bone marrow, where it effectively delivers mRNA to a broad spectrum of immune cells, including macrophages, dendritic cells, T cells, B cells, and NK cells. This capability enables its core application: the in vivogeneration of "pan-CAR" immune cells. When loaded with anti-HER2 CAR mRNA, the H5T5-based therapy achieved potent tumor regression and prolonged survival in multiple solid tumor models. Preliminary safety assessments indicated a manageable cytokine profile and no significant organ toxicity, positioning it as a promising platform for in vivocell engineering.
More description
|
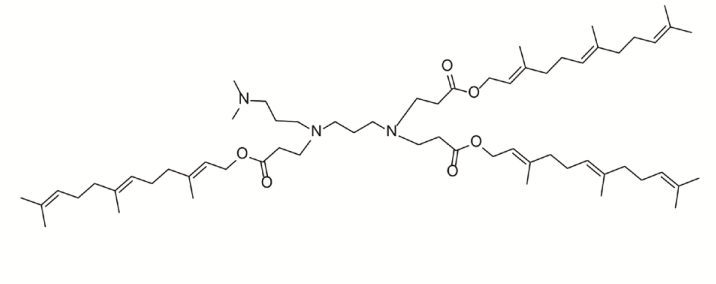
|
| DC67556 | Sail Lipid 2308 Featured |
Sail Lipid 2308 is a novel ionizable lipid targeting to spleen developed by Sai Biomedicine.As described on US20250205167A1, Lipid 2308 was designed with a piperidine core (6-membered ring) and asymmetric C17/C11 chains, this lipid achieves unprecedented spleen-specificity. It demonstrates dominant spleen accumulation (Spleen RLU: 7.8E+06, 91.8% of total signal) with a record spleen-to-liver ratio of 112.7 (9× higher than 2231). Despite lower protein expression (hEPO: 11,000 ng/mL), near-zero liver uptake (Liver RLU: 66,000) makes Lipid 2308 unparalleled for vaccine/immunotherapy applications targeting splenic immune cells.
More description
|
|
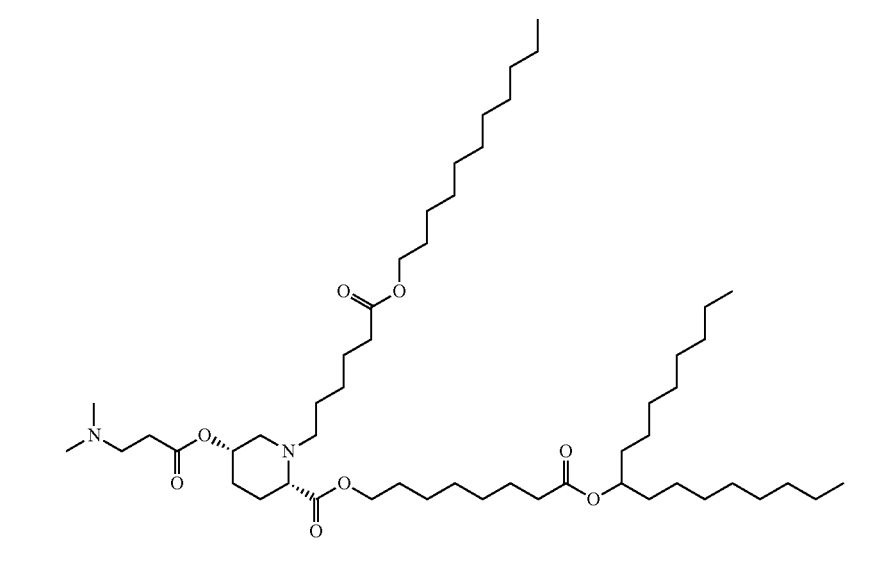
| DC67652 | CICL-242 Featured |
CICL-242 is a constrained ionizable cationic lipid highlighted in patent US 20250127728A1 developed by Capstan as a promising candidate for advanced therapeutic delivery, particularly in stem cell and gene editing applications. Its structure features a rigid amine headgroup similar to CICL-207, which likely facilitates efficient endosomal escape and reduces non-specific uptake, enhancing targeted nucleic acid delivery. Although detailed performance data is not fully disclosed in the patent, CICL-242 is explicitly synthesized and included in gene editing experimental systems (e.g., CRISPR-Cas9 workflows), suggesting its potential for high-efficiency transfection in hard-to-transfect cells like hematopoietic stem cells (CD34⁺). This makes it a strong candidate for ex vivo cell engineering and regenerative medicine, where precision and low off-target effects are critical. While further validation is needed to quantify its efficacy and safety profile, CICL-242 represents a strategic innovation in the lipid library for next-generation genetic therapies.
More description
|
|
| DC60880 | 2Ac3-C18 Featured |
2Ac3-C18 is a unique ionizable lipid with a distinct degradable core structure:featuring 2 acrylate units and 3 amine groups—linked to a C18 alkyl chain. Its LNPs (formulated with DOPE/cholesterol/DMG-PEG2000) exhibit spleen-specific mRNA delivery in vivo.
More description
|

|
| DC67633 | Lipid KEL12 |
(4S)-KEL12 is a novel, biodegradable ionizable lipid developed for advanced mRNA vaccine delivery. It was rationally designed by incorporating both a ketal group in the linker and ester segments in the hydrophobic tails, a dual-degradable strategy aimed at enhancing its safety profile. Through iterative optimization, (4S)-KEL12 was identified as a lead candidate with an optimal pKa value of approximately 6.78, which is crucial for efficient mRNA encapsulation and endosomal release.
More description
|
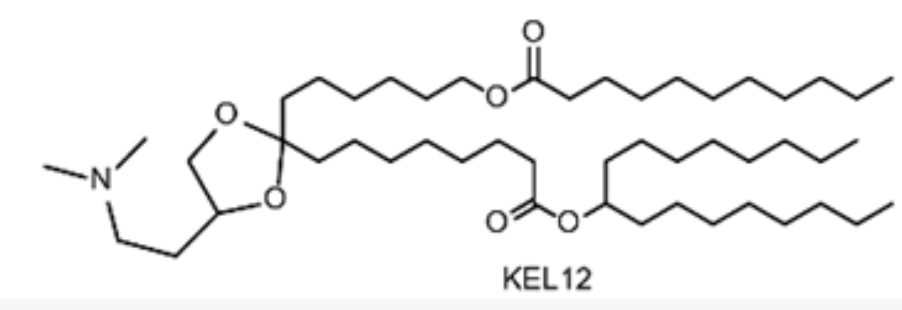
|
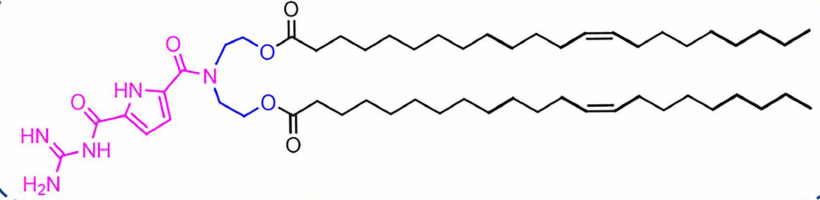
| DC67632 | Lipid GL5 |
GL5 is an ionizable guanidine-based lipid nanoparticle (G-LNP) designed for superior mRNA delivery. Its guanidinocarbonyl-pyrrole (GCP) headgroup enables pH-responsive behavior and strong mRNA binding via bidentate hydrogen bonds. The cholesterol-free GL5-3 formulation forms compact, stable nanoparticles (~90-120 nm) that exhibit excellent spleen-targeting capability after intravenous injection.GL5-LNPs efficiently deliver mRNA to antigen-presenting cells (APCs), enhancing antigen presentation and T cell activation. In cancer immunotherapy models, GL5-based mRNA vaccines provided complete tumor protection and induced durable immune memory. The platform also enables mRNA delivery to other organs like the pancreas via different administration routes, demonstrating remarkable versatility and therapeutic potential.
More description
|
|
| DC67651 | CICL-238 |
Based on the data from patent US 20250127728A1, CICL-238 emerges as a highly promising ionizable lipid candidate, demonstrating notable advantages for targeted delivery applications. It achieves exceptional transfection efficiency—reaching approximately 90% of CICL-207's performance in splenic T-cells even at a reduced lipid ratio of 50% in LNP formulations. Additionally, CICL-238 exhibits minimal off-target expression in hepatocytes (<8%, comparable to CICL-207), underscoring its enhanced specificity for immune cells over liver tissues. Its optimized structure likely contributes to efficient endosomal escape and reduced Kupffer cell uptake, making it ideal for liver-related therapies (e.g., siRNA silencing for metabolic diseases) and potentially broadening applications to genetic medicine where precision and safety are paramount. Further validation in disease models could solidify its role as a versatile, low-toxicity alternative to benchmark lipids.
More description
|
|
| DC67995 | Lipid 22 Featured |
Compound 22, as detailed in United States Patent US 2026/0014089 A1, is a bifunctional ionizable lipid engineered for precision drug delivery. Its structure integrates a monosaccharide targeting headgroup, designed to bind specifically to DC-SIGN receptors on dendritic cells, via a sophisticated linker connected to a biodegradable lipid anchor. This design enables it to serve as a key component of lipid nanoparticles (LNPs), forming a targeted delivery system. By leveraging the specific carbohydrate-receptor interaction, these LNPs are preferentially internalized by dendritic cells, critical for initiating adaptive immune responses. In vivo studies from the patent, such as the biodistribution data shown in Figure 5, confirm effective accumulation in lymphoid tissues like the spleen and lymph nodes. Consequently, this targeted delivery enhances the potency of encapsulated payloads (e.g., mRNA vaccines) by ensuring professional antigen presentation, eliciting a stronger and more specific immune response—evidenced by higher neutralizing antibody titers—making it a powerful tool for next-generation vaccines and therapeutics.
More description
|

|
| DC67549 | ORNA lipid 144(1-C) |
ORNA lipid 144 is a novel ionizable lipid engineered for splenic RNA delivery developed by ORNA Therapeutics, featuring a biodegradable structure with a protonatable tertiary amine headgroup and ester-linked branched C14 alkyl chains. This design enables exceptional spleen-targeting capability, demonstrated by 3-fold higher luciferase expression in the spleen compared to benchmark lipids and near-complete B-cell depletion when delivering anti-CD19 CAR circRNA. It forms highly stable lipid nanoparticles maintaining homogeneous size (60–80 nm) and low polydispersity across diverse manufacturing conditions and buffer systems. Rapid clearance from the liver and spleen minimizes off-target accumulation, while high circRNA encapsulation efficiency (>90%) and pH-dependent endosomal escape make it ideal for immunotherapies and vaccines requiring precise splenic bioavailability and sustained efficacy.
More description
|
|
| DC67460 | CP-LC-1422 |
Derived from the natural amino acid homocysteine, CP-LC-1422 is an ionizable cationic lipid that enables robust in vivo delivery of various RNA forms (mRNA, cRNA, and saRNA), driving high protein expression. When formulated into LNPs (50/38.5/10/1.5 molar ratio of ionizable lipid/cholesterol/DOPE/PEG-lipid), it achieves superior spleen-specific targeting compared to commercial options through intravenous administration, while maintaining an excellent safety profile.
More description
|

|
| DC67281 | BNT-51 Featured |
BNT-51 is an ionizable thiolipid developed by Biontech, characterized by its sulfur-containing moieties and a multiarm dendron-like architecture. Synthesized via reactions between amine-containing compounds and sulfur-based halides or sulfonates, it forms stable lipid nanoparticles (LNPs) optimized for mRNA delivery. The LNPs exhibit uniform particle size (80–100 nm, PDI <0.2), near-neutral zeta potential, and high mRNA encapsulation efficiency (>90%), while maintaining payload integrity through freeze-thaw cycles and extended storage. In vitro, BNT-51 demonstrates low cytotoxicity (>80% cell viability in C2C12, HepG2, and HEK293 cells) and superior transfection efficiency compared to conventional lipids, particularly in immune cells such as CD4+/CD8+ T cells within PBMCs. Its modular design allows integration of stealth lipids (e.g., PEG or vitamin E derivatives) to prolong circulation time and minimize immune activation, as evidenced by low hemolysis and complement activation risks. In vivo, BNT-51-based LNPs enable targeted mRNA delivery to splenic macrophages, achieving potent genome editing (e.g., Cre mRNA) and therapeutic protein expression (e.g., BACH1) in preclinical models. With its tunable structure, robust stability, and cell-specific tropism, BNT-51 holds promise for advancing mRNA therapeutics in gene editing, cancer immunotherapy, and regenerative medicine, offering a versatile platform for next-generation nanomedicine.
More description
|
|
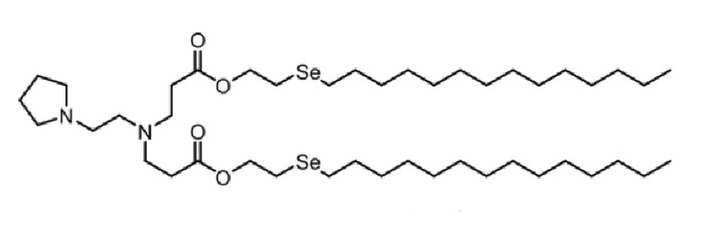
| DC60494 | 76-O17Se |
76-O17Se is a lipidoid for the efficient delivery of antiCD19 mRNA CAR to murine primary macrophages. 76-O17Se is more efficient than delivery with lipofectamine 2000 (LPF2K) or MC3
More description
|
|
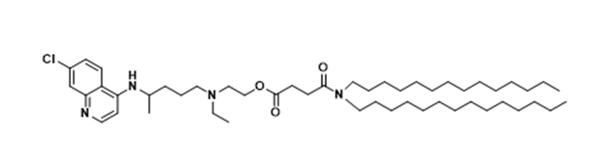
| DC67544 | HCQ Lipid 4(HCQ-4) |
HCQ-4 is a rationally engineered ionizable lipid derived from hydroxychloroquine (HCQ), featuring a ditetradecylamine-derived twin-C14 saturated hydrocarbon tail linked to the HCQ headgroup via a succinic acid spacer. Synthesized through a three-step route involving HCQ deprotonation, ditetradecylamine carboxylation, and EDC/DMAP-mediated amidation, this lipid forms the core of optimized lipid nanoparticles (LNPs) at a molar ratio of 60:10:40:0.5 (HCQ-4:DOPE:cholesterol:DMG PEG2000). The structure enables dual functionality: (1) Spleen-selective mRNA delivery (2.3-fold higher splenic vs. hepatic transfection) via 80-100 nm particle size, near-neutral charge (-3 mV), and low PEG density, facilitating immune cell uptake; (2) Tumor microenvironment modulation through HCQ-mediated repolarization of M2 macrophages to antitumor M1 phenotype (iNOS+ cells ↑2.5-fold, CD206+ cells ↓60%). This bifunctional design synergistically enhances mRNA cancer vaccine efficacy, demonstrating superior prophylactic/therapeutic antitumor activity and antimetastatic effects compared to clinical benchmarks like MC-3 LNP.
More description
|
|
| DC60537 | C18 NC-TNP Featured |
NC-TNP (noncationic thiourea lipids nanoparticles) could compress mRNA by strong hydrogen bonds interaction between thiourea groups of NC-TNP and the phosphate groups of mRNA. NC-TNP could escape the recycling pathway to inhibit the egress of internalized nanoparticles from the intracellular compartment to the extracellular milieu. NC-TNP-encapsulated mRNA shows higher gene transfection efficiency in vitro and in vivo than mRNA-LNP formulation. NC-TNP also shows spleen targeting delivery ability with higher accumulation ratio (spleen/liver), compared with traditional LNP.The C18 non-cationic thiourea lipid self-assembles into ~100 nm nanoparticles with neutral surface charge, utilizing strong hydrogen bonding between its thiourea groups and mRNA phosphate groups for efficient mRNA complexation. This delivery system demonstrates significantly enhanced EGFP expression efficiency—2.3-fold higher than standard C6/C12 formulations—in DC2.4, B16, and 4T1 cells, while sustaining luciferase activity for over 20 days post-subcutaneous injection. It exhibits exceptional stability, maintaining >94% mRNA integrity and <10% particle size variation after 30-day lyophilized storage. Importantly, the nanoparticles show pronounced spleen-targeting capability with 20-fold greater accumulation in the spleen versus liver, effectively activating twice the level of antigen-specific CD8⁺ T cells. Critically, the system avoids cationic lipid-associated toxicity, inducing no detectable IL-6/CXCL10 inflammation and causing no histopathological damage in cardiac or splenic tissues, thus establishing a novel high-efficacy, low-toxicity mRNA delivery platform.
More description
|

|
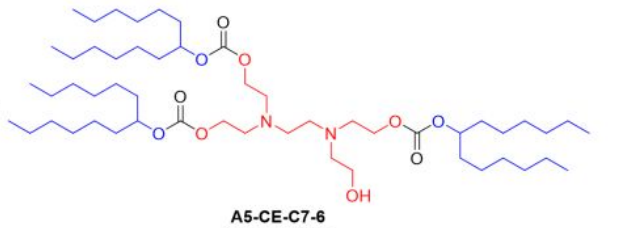
| DC67540 | Lipid A5-CE-C7-6 |
A5-CE-C7-6 is an ionizable lipid engineered for spleen-targeted mRNA delivery, integrating a hydroxylated dual-amine core (A5) for enhanced mRNA binding and endosomal escape, a biodegradable carbonate ester linker (CE) enabling rapid hydrolysis (61% degradation in 24 h), and branched heptyl hydrophobic tails (C7-6) that optimize nanoparticle stability and spleen tropism. When formulated into cholesterol-free lipid nanoparticles (B-8 formulation), its unique architecture—combining hydroxyl groups for cellular uptake, carbonate-mediated biodegradability, and branched-chain fluidity—achieves unprecedented efficiency: low pKa (~6.0) minimizes liver accumulation while enabling 21% transfection of splenic NK cells, outperforming benchmark systems like MC3 SORT LNPs by >10-fold in spleen-specific delivery and establishing a new standard for in vivo immune cell engineering.
More description
|
|
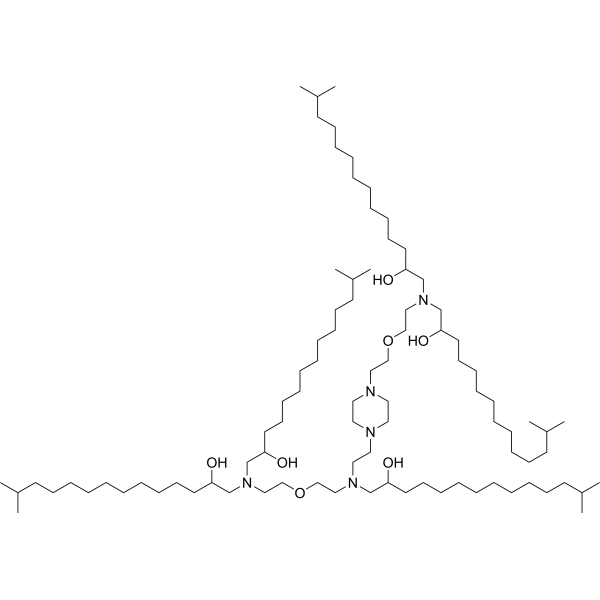
| DC13101 | E10i-494 Featured |
E10i-494 is a branched ionizable lipid designed to enhance the delivery of mRNA and CRISPR-Cas9 ribonucleoprotein (RNP) complexes. It belongs to the Branched Endosomal Disruptor (BEND) lipid family, which features terminal branching to improve endosomal escape and cellular uptake.E10i-494 demonstrated exceptional performance in T cell engineering, achieving >80% transfection efficiency in primary human T cells. This is significantly higher than the ~70% efficiency achieved by the linear lipid C14-494.The isopropyl branch enhances the lipid's ability to penetrate and disrupt endosomal membranes, leading to improved release of mRNA and RNPs into the cytoplasm.Despite its high efficiency, E10i-494 exhibits low cytotoxicity, making it suitable for therapeutic applications.E10i-494 is particularly effective for delivering mRNA to T cells, making it a promising tool for CAR-T cell therapy and other immunotherapies.Its ability to deliver CRISPR-Cas9 RNPs efficiently also makes it suitable for in vivo gene editing applications.
More description
|
|
| DC67565 | IAJD249 |
IAJD 294 is a single-component ionizable amphiphilic Janus dendrimer that autonomously coassembles with mRNA via simple injection into uniform monodisperse dendrimersome nanoparticles (DNPs, 85 nm diameter, PDI<0.2), eliminating complex multi-component formulations. Its optimized 3,5-benzoyl ester linkage and symmetric hydrophobic tails enable dual-organ targeting:
Spleen: 2.97 × 10⁷ RLU (50% of total activity)
Lymph nodes: 10⁶ RLU (10× higher than IAJD 87)
through partial hydrophobic interdigitation (stabilizing DNPs for enhanced lymphatic uptake) and pKa ~6.5 (facilitating endosomal escape), validating constitutional isomerism for precision delivery.
More description
|
|
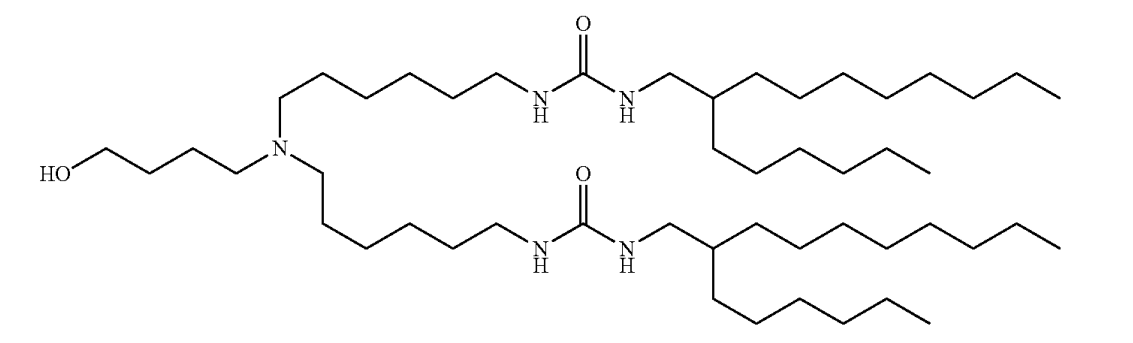
| DC67517 | Westgene lipid 8 |
Westgene lipid 8 is a cationic lipid featuring a tertiary amine core with three alkyl chains (C1-C15) and two unsaturated C18 linoleate-like tails. Its ionizable amine enables pH-dependent charge for mRNA encapsulation in LNPs. Key structural elements include branched alkyl groups (X1/X2: C4, X3: C2) and ester-linked unsaturated R1/R2 chains, enhancing membrane fusion and endosomal escape. N Used in lipid nanoparticles (LNPs) with DOPE, cholesterol, and PEG-DMG, it demonstrates low cytotoxicity, high mRNA delivery efficiency, and spleen-targeted immune activation, making it suitable for vaccine/therapeutic delivery.
More description
|
|
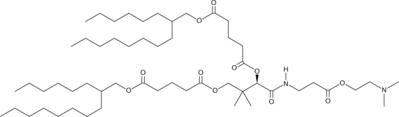
| DC60684 | Lipid I97 |
Lipid I97 is a vitamin B5-derived ionizable lipid for mRNA vaccine delivery. Lipid I97 LNP specifically delivers the mRNA to the spleen and lymph nodes in model mice, induces balanced Th1/Th2 immune responses, and elicits the production of high levels of neutralizing antibodies with low toxicity.
More description
|
|
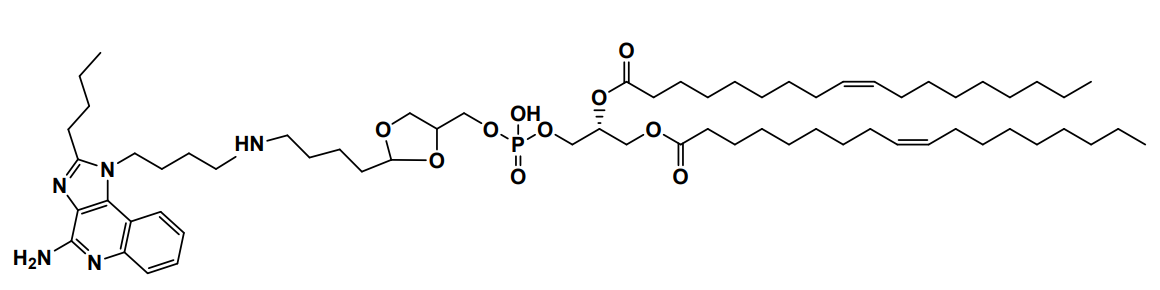
| DC60828 | YK-TLR-001 Featured |
YK-TLR-001 is a cyclic acetal-based ionizable lipid for mRNA delivery. YK-TLR-001 LNPs are demonstrated to enhance mRNA expression in the spleens and to induce exceptional maturation of antigen-presenting cells (APCs) and to promote antigen presentation.
More description
|
|